PRADALEX™ (pradofloxacin injection)
Bovine respiratory disease (BRD) and Swine respiratory disease (SRD) remain a major challenge, even as management practices and interventions continue to improve. Pradalex™ brings a novel mode of action with a hard-hitting bactericidal efficacy against the four labeled BRD pathogens, M. haemolytica, P. multocida, H. somni, and M. bovis, and the five labeled SRD pathogens, Bordetella bronchiseptica, Glaesserella (Haemophilus) parasuis, Pasteurella multocida, Streptococcus suis, and Mycoplasma hyopneumoniae.
SIGNATURE CHARACTERISTICS

Pradofloxacin is an innovative, third-generation fluoroquinolone
Blocks two enzymes responsible for bacterial replication with equal affinity in the same organism

Convenient single-dose, low-volume injection
PRADALEX IS READY TO TAKE ON:
after consideration of other non-fluoroquinolones therapeutic options.
BRD BACTERIA

Histophilus somni

Mannheimia haemolytica
Mycoplasma bovis

Pasteurella multocida
SRD BACTERIA

Bordetella bronchiseptica

Glaesserella (Haemophilus) parasuis

Pasteurella multocida

Streptococcus suis

Mycoplasma hyopneumoniae
PRADALEX IN ACTION
Fluoroquinolone antibiotics work by binding and inactivating DNA gyrase and topoisomerase IV. This inhibits bacterial replication and causes the chromosomes to fragment into pieces — resulting in cell death. Most fluoroquinolones act primarily on DNA gyrase but Pradalex is dual acting.
Pradalex has an equal affinity to both DNA gyrase and topoisomerase IV, making it a potent fluoroquinolone.
Pradalex is ready to go to work for your operation.
POWERFUL PUNCH, SMALL DOSE
Pradalex brings the benefits without compromising on delivery. This antibiotic is a one-shot, low-volume injection — with excellent syringeability.2
Pradalex is rapidly absorbed and is cleared quickly with an industry-leading withdrawal period.
Hard-hitting against bacteria. Easy on your production teams. Pradalex is designed to be a treatment to fight BRD and SRD only after consideration of other non-fluoroquinolone therapeutic options.
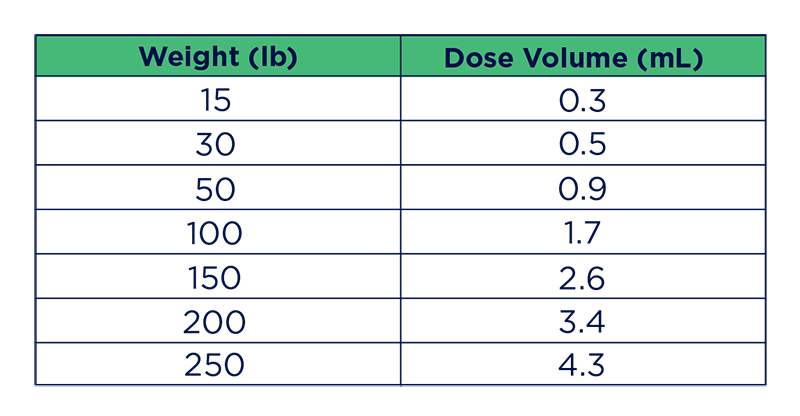
Pradalex Dosing Guide for Swine (1.7 mL/100 lbs)
Talk to your Elanco team today about Pradalex product availability and see how Pradalex might help combat respiratory disease in your operation.
RESPIRATORY SOLUTIONS
The Elanco Portfolio of products offers immune support, preventative measures, and quick interventions against respiratory disease are essential to not only your herd’s health but to the health of your bottom line. Protect your investment with Elanco to address common respiratory disease challenges.


Sign up to stay informed about Pradalex and Elanco’s line of respiratory solutions.
The labels contain complete use information, including cautions and warnings. Always read, follow and understand the label and use directions.
Pradalex™
INDICATIONS:
Cattle: Pradalex is indicated for the treatment of bovine respiratory disease (BRD) associated with Mannheimia haemolytica, Pasteurella multocida, Histophilus somni and Mycoplasma bovis in cattle intended for slaughter (beef calves 2 months of age and older, growing beef steers, growing beef heifers, and beef bulls intended for slaughter), and in cattle intended for breeding less than 1 year of age (replacement beef and dairy heifers less than 1 year of age and beef and dairy bulls less than 1 year of age). Not for use in cattle intended for breeding 1 year of age and older (replacement beef and dairy heifers 1 year of age and older, beef and dairy bulls 1 year of age and older, and beef and dairy cows), beef calves less than 2 months of age, dairy calves, and veal calves.
Swine: Pradalex is indicated for the treatment of swine respiratory disease (SRD) associated with Bordetella bronchiseptica, Glaesserella (Haemophilus) parasuis, Pasteurella multocida, Streptococcus suis and Mycoplasma hyopneumoniae in weaned swine intended for slaughter (nursery, growing and finishing swine, boars intended for slaughter, barrows, gilts intended for slaughter, and sows intended for slaughter.) Not for use in swine intended for breeding (boars intended for breeding, replacement gilts, and sows intended for breeding) and in nursing piglets.
IMPORTANT SAFETY INFORMATION:
CAUTION: Federal law restricts this drug to use by or on the order of a licensed veterinarian and prohibits the extra-label use of this drug in food-producing animals. To ensure responsible antimicrobial drug use, use of pradofloxacin should be limited to treatment of bovine respiratory disease (BRD) in cattle and swine respiratory disease (SRD) in swine only after consideration of other non-fluoroquinolone therapeutic options.
Not for use in humans. Keep out of reach of children. Avoid contact with eyes and skin. Individuals with a history of hypersensitivity to quinolones should avoid this product. Not for use in animals intended for breeding. Not for use in beef calves less than 2 months of age, dairy calves, veal calves and nursing piglets; a withdrawal period has not been established for this product in pre-ruminating calves. Quinolones should be used with caution in animals with known or suspected central nervous system (CNS) disorders. Mild to moderate inflammatory changes of the injection site may be seen in animals treated with Pradalex. Pradalex has a pre-slaughter withdrawal time of 4 days for cattle and 2 days for swine. See package insert for additional safety information.
